
 Document
Document
The Role of a Biosynthetic Hybrid Mesh in Abdominal Wall Hernia Repair in High-Risk Patients with Multiple Comorbidities
The influx of more high-risk and obese patients in need of abdominal wall hernia repair naturally leads to larger, more complex hernia cases and the need for strong mesh.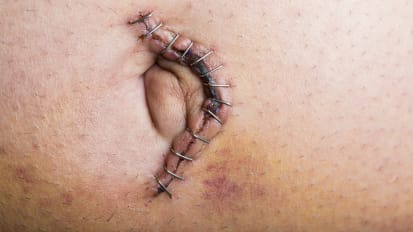 Document
Document
Clinical Performance with Staple Line Reinforcement: Scientific Literature Analysis
Clinical Performance of GORE® SEAMGUARD® Bioabsorbable Staple Line Reinforcement. Document
Document
20 Years of Clinical History: Minimizing Complications, Maximizing Outcomes
Partner with a proven leader in reducing risks and raising the standard for bariatric and hiatal / paraesophageal hernia surgery. Document
Document
Clinical Use of GORE® BIO-A® Tissue Reinforcement in Ventral Hernia Repair Using the Components Separation Technique
Many repaired incisional or ventral hernias recur; thus, the primary objective of research and technological developments pertaining to these hernias has long been to minimize recurrences without increasing treatment-related complications. Document
Document
Expert Review: Clinical experience with GORE® SYNECOR Intraperitoneal Biomaterial
A surgeon reviewing their clinical experience with GORE® SYNECOR Intraperitoneal Biomaterial and MEDTRONIC PARIETEX Composite Mesh for Umbilical / Epigastric Hernias. Video
Video
Laparoscopic Fixation of GORE® SYNECOR Biomaterial in a Cadaver Model Using a Robotic Arm
Erik B. Wilson M.D., FACS, shares a video showing laparoscopic fixation of GORE® SYNECOR Biomaterial in a cadaver model using a robotic arm. Document
Document
Case Study: Proven Outcomes Reduced Costs for Complex Hernia Repairs
GORE® BIO-A® Tissue Reinforcement is an excellent choice in soft tissue repair, including complex hernia.